Abstract
Background:
The efficacy of efSITT with beclometasone dipropionate/formoterol fumarate/glycopyrronium (BDP/FF/G) in asthma has been assessed in randomized controlled trials excluding though both current and ex-smokers ≥ 10 pack years¹. Triangle provides real-world evidence of efSITT's effectiveness in asthma patients in Greece. Aims and objectives: This observational study aims to assess therapy pathways and health-related outcomes of BDP/FF/G, including patients of all smoking statuses.
Methods:
This analysis includes 333 patients who received treatment with BDP/FF/G (87/5/9 µg) for 1 year. The co-primary objective is to evaluate asthma control (ACT) and in this interim analysis results were stratified by patients’ smoking status.
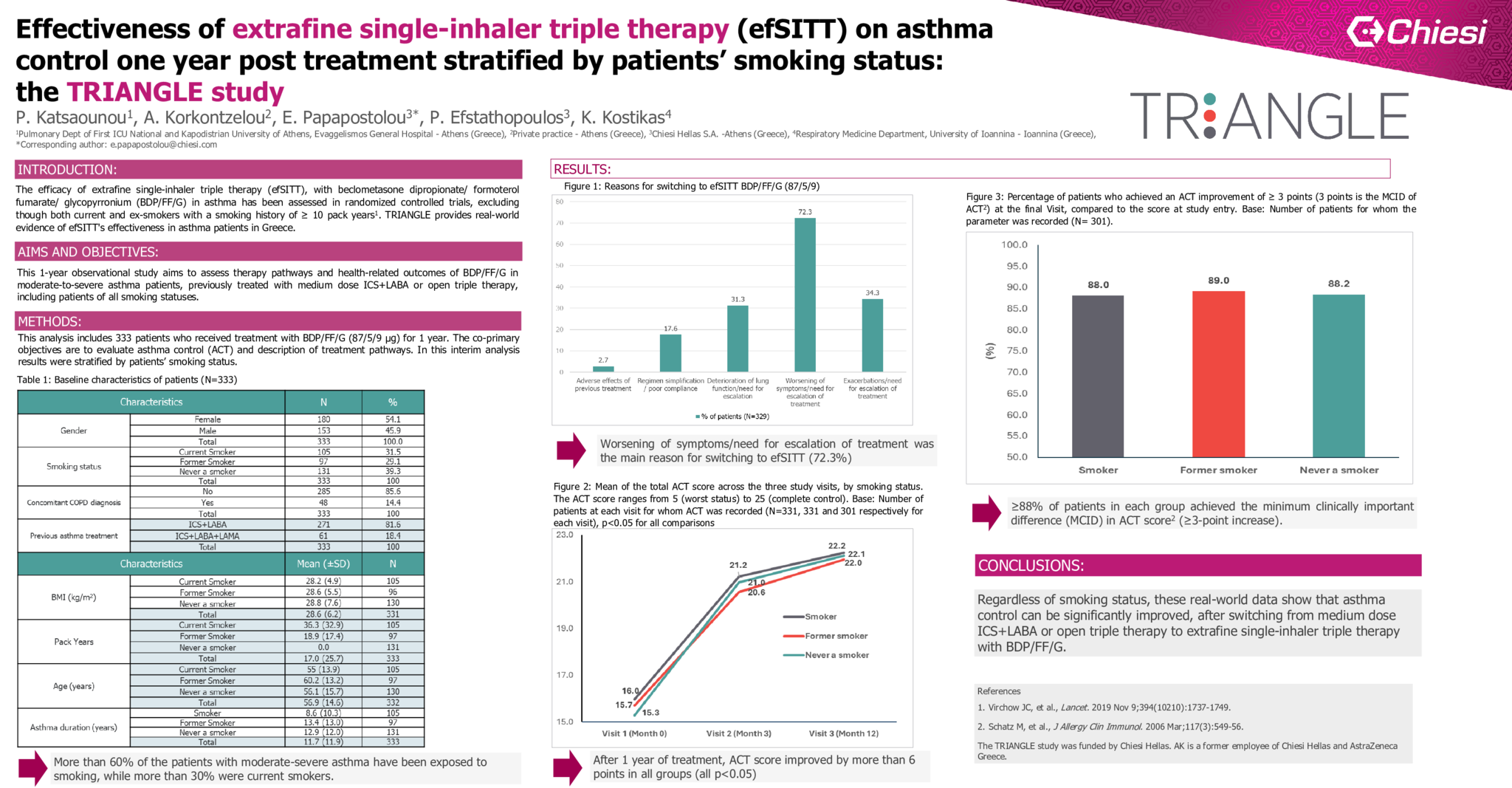
Results:
Overall, 39.3% of patients were never smokers, 31.5% were current smokers and 29.1% ex-smokers with a mean history of 36.3 and 18.9 pack years, respectively. After 1 year of treatment, ACT score improved by more than 6 points (pts) in all groups (p<0.05) (Figure 1A). The percentage of patients who achieved an ACT improvement of ≥ 3 pts reached or exceeded 88% in all three groups (Figure 1B).
Conclusions:
Our real-world data suggest a significant improvement in asthma control after switching to BDP/FF/G regardless of smoking status, enhancing our understanding on efSITT's effectiveness in these understudied populations.
This study was funded by Chiesi Hellas S.A.
- 3 προβολές